Sharps Disposal in Dental Practices: Best Practices for Safety
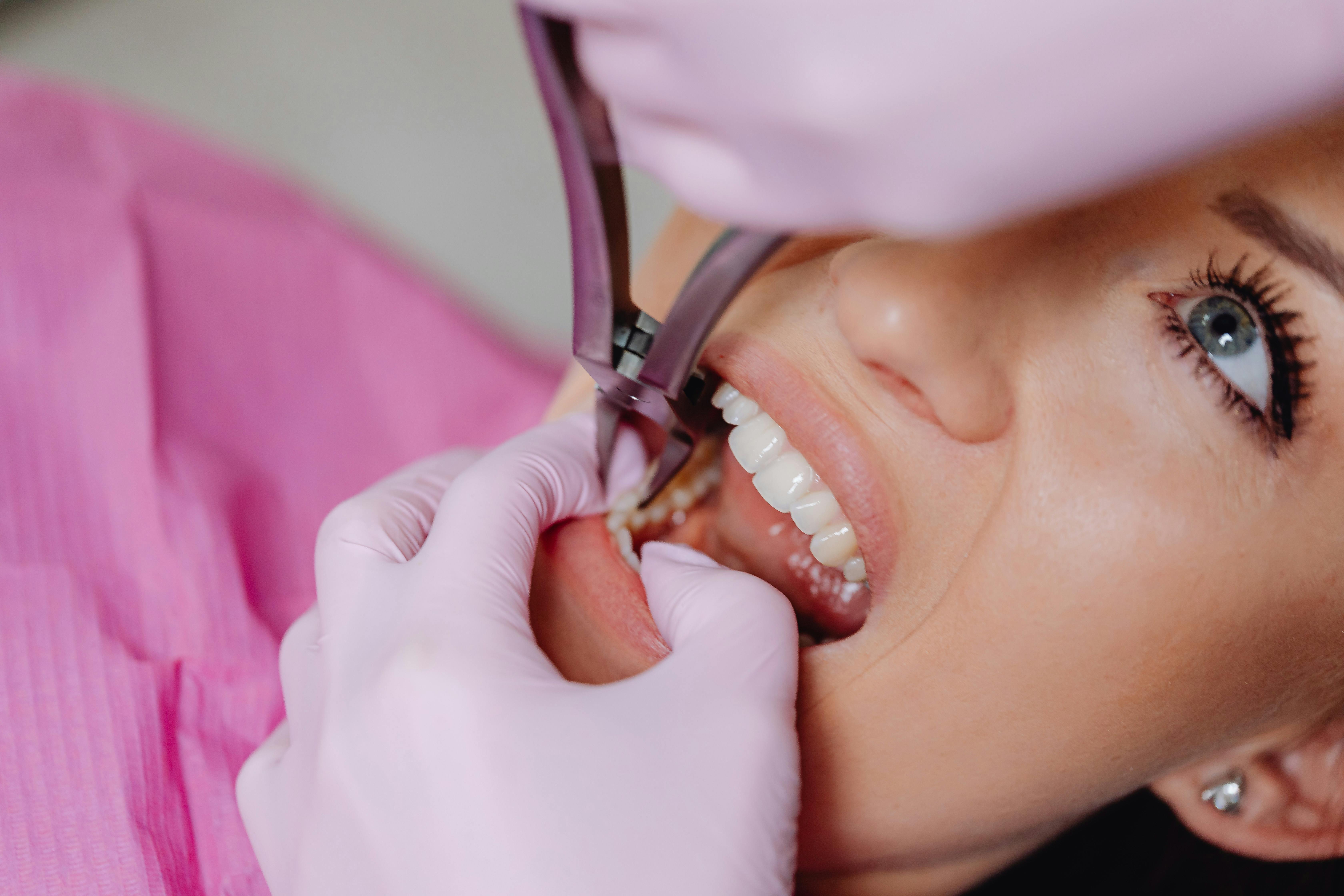
Sharps injuries remain one of the most significant occupational hazards in dental practice. Every year, thousands of dental professionals across the United Kingdom sustain needlestick injuries or cuts from contaminated sharp instruments, exposing them to potentially life-threatening blood-borne viruses including Hepatitis B, Hepatitis C, and HIV. The safe disposal of sharps is not merely a matter of good housekeeping — it is a fundamental legal obligation, a cornerstone of infection control, and a direct reflection of how seriously a practice takes the welfare of its team. In this comprehensive guide, we examine every aspect of sharps disposal in the dental setting, from the regulatory framework that governs it to the practical day-to-day procedures that keep your staff and patients safe.
What Counts as a "Sharp" in Dentistry?
Before establishing safe disposal protocols, it is essential that every member of the dental team understands precisely what constitutes a sharp. The definition extends far beyond the obvious hypodermic needle. In dentistry, a sharp is any item that could cause a penetrating injury and may be contaminated with blood, saliva, or other potentially infectious material.
Common Sharps Found in Dental Practice
- Hypodermic needles — Used for administering local anaesthesia, these are the most commonly encountered sharps in general dental practice. Both short and long needles present identical disposal requirements.
- Anaesthetic cartridges — Glass cartridges, whether intact or broken, are classified as sharps due to the risk of glass penetration and potential blood contamination from needle re-entry.
- Scalpel blades — Used in oral surgery, periodontal procedures, and some endodontic treatments. These must never be removed from handles by hand without an appropriate removal device.
- Dental burs — Both diamond and tungsten carbide burs become contaminated during use and their sharp cutting surfaces can easily penetrate skin and gloves.
- Orthodontic wires — Archwires, ligature wires, and sectional wires have sharp cut ends that can cause puncture injuries during handling and disposal.
- Matrix bands — Metal matrix bands, particularly Tofflemire and sectional matrices, have thin sharp edges that can lacerate skin.
- Endodontic files and reamers — These fine instruments are easily capable of penetrating gloves and skin, and are frequently contaminated with pulpal blood.
- Irrigation needles — Side-venting and conventional irrigation needles used in endodontic procedures require sharps disposal.
- Broken instruments — Any instrument that has fractured during use, creating a sharp edge or point, must be treated as a sharp.
- Suture needles — Used in oral surgery and periodontal procedures, these curved needles are a common source of sharps injuries.
- Disposable explorers and probes — Single-use sharp diagnostic instruments must be disposed of as sharps rather than in general clinical waste.
- Ultrasonic scaler tips — While not always considered, damaged or single-use scaler tips with sharp points qualify as sharps.
Key Principle
If an item can penetrate the skin and has been in contact with blood or saliva, it must be disposed of as a sharp. When in doubt, always err on the side of caution and place the item in a sharps container. This protects not only your clinical team but also cleaning staff, waste handlers, and the wider community.
Understanding this broad definition is particularly important for training purposes. Dental nurses, hygienists, and therapists must all recognise that sharps disposal extends well beyond simply placing used needles in a yellow bin. A comprehensive approach to sharps disposal acknowledges every potential source of penetrating injury within the practice.
The Legal Framework Governing Sharps Disposal in Dental Practice
Dental practices in England operate within a complex web of legislation and guidance relating to sharps safety. Understanding these requirements is essential for compliance and, more importantly, for the protection of your team. Non-compliance can result in enforcement action, significant fines, and in the most serious cases, criminal prosecution.
Health and Safety (Sharp Instruments in Healthcare) Regulations 2013
These regulations, which came into force on 11 May 2013, implement the EU Council Directive 2010/32/EU in the United Kingdom. They place specific duties on employers in the healthcare sector to protect workers from injuries caused by sharp instruments. The key requirements include:
- Risk assessment — Employers must carry out a specific assessment of the risks from sharps injuries, going beyond the general risk assessment requirements of the Management of Health and Safety at Work Regulations 1999.
- Elimination and substitution — Where it is reasonably practicable, the use of sharps must be eliminated entirely. Where this is not possible, safer sharps devices must be substituted for conventional instruments.
- Safe use procedures — Employers must establish and implement safe procedures for using and disposing of sharps, including an absolute prohibition on the recapping of needles unless a risk assessment demonstrates that recapping is itself the safest option.
- Information and training — All workers who may be exposed to sharps must receive adequate training on the risks, preventive measures, reporting procedures, and response protocols.
- Investigation and response — Every sharps injury must be investigated to determine its cause and to identify measures to prevent recurrence.
HTM 07-01: Safe Management of Healthcare Waste
Health Technical Memorandum 07-01 provides the definitive guidance on waste management in healthcare settings, including dental practices. It establishes the waste classification system, specifies container requirements, and sets out the procedures for segregation, storage, and collection. For sharps specifically, HTM 07-01 confirms that all sharps contaminated with medicinal products must be placed in yellow-lidded sharps containers, while sharps not contaminated with medicines (but contaminated with blood or body fluids) should also be placed in yellow-lidded containers for incineration.
Control of Substances Hazardous to Health (COSHH) Regulations 2002
COSHH applies to sharps disposal because biological agents (blood-borne viruses) constitute substances hazardous to health. The regulations require employers to assess the risk from biological agents, implement control measures, and provide health surveillance where appropriate. In the context of sharps, this means treating all blood and saliva as potentially infectious and implementing universal precautions.
Environmental Protection Act 1990 and Duty of Care
Under the Environmental Protection Act 1990, dental practices have a duty of care regarding all waste they produce. This means ensuring that sharps waste is stored safely, described accurately on waste transfer documentation, transferred only to authorised persons, and that the practice retains consignment notes for a minimum of three years. Failure to comply with duty of care requirements is a criminal offence that can result in unlimited fines.
CQC Regulation 12: Safe Care and Treatment
The Care Quality Commission enforces Regulation 12 of the Health and Social Care Act 2008 (Regulated Activities) Regulations 2014, which requires providers to assess and mitigate risks to health and safety, including those arising from sharps. CQC inspectors specifically examine sharps management as part of their assessment of the "Safe" domain. For more details on how to ensure your practice meets all regulatory requirements, visit our compliance information page.
Regulatory Warning
Since 2013, the use of safer sharps devices has been a legal requirement wherever reasonably practicable. CQC has explicitly stated that practices should be able to demonstrate why they have chosen specific devices and show evidence that safer alternatives have been considered. Simply continuing to use conventional needles without documented justification is a compliance failure.
Choosing the Right Sharps Container for Your Dental Practice
The selection of appropriate sharps containers is a critical decision that affects safety, compliance, and cost-effectiveness. Not all sharps containers are created equal, and dental practices have specific requirements that differ from other healthcare settings.
Standards and Certification
All sharps containers used in UK dental practices must comply with BS 7320:1990 (the British Standard for sharps containers) and be UN3291 approved for the transport of clinical waste. These standards ensure that containers are puncture-resistant, leak-proof, and capable of withstanding the rigours of transportation to treatment facilities. When selecting containers, always verify that they carry the appropriate markings confirming compliance with these standards.
Colour Coding
In accordance with HTM 07-01, dental practices should use yellow containers with yellow lids for sharps contaminated with medicines (which includes local anaesthetic needles and cartridges). This colour coding indicates that the waste requires incineration. The colour system is not merely advisory — it is essential for ensuring that waste follows the correct treatment pathway. Incorrect colour coding can result in waste being sent to inappropriate treatment facilities, creating regulatory and environmental problems.
Size Selection
Sharps containers are available in a range of sizes, and selecting the appropriate size for each location in your practice is important for both safety and efficiency:
- 0.5 litre containers — Suitable for individual treatment rooms with low sharps generation, or as a secondary container for specific procedures.
- 1 litre containers — The most common choice for individual dental surgeries. Compact enough to sit on a bracket table or worktop without taking up excessive space, yet large enough to last a reasonable period before requiring replacement.
- 2.5 litre containers — Appropriate for busy surgeries or practices where multiple procedures generating sharps are performed in the same room.
- 5 litre containers — Suitable for oral surgery suites or as a central collection point where smaller containers are emptied (note: decanting sharps between containers is not recommended).
- 7 litre and above — Rarely needed in individual treatment rooms but may be appropriate for central storage areas in larger practices or community dental clinics.
Best Practice Tip
Each treatment room should have its own dedicated sharps container, positioned within arm's reach of the operator. The container should be mounted securely at a height between waist and shoulder level, ensuring that sharps can be deposited without reaching over other items or across the patient. Wall-mounted brackets provide the most secure and accessible positioning.
Container Features to Look For
Modern sharps containers offer several features that enhance safety in dental practice:
- Temporary closure mechanism — Allows the container to be partially closed when not in active use, preventing accidental spillage or unauthorised access.
- Wide aperture design — Facilitates safe single-handed disposal of needles still attached to syringes, reducing the risk associated with needle removal.
- Integral needle removal feature — Some containers incorporate a built-in notch for safely detaching needles from syringes without hand involvement.
- Clear fill-level indicator — A visible line marking the maximum fill level (typically three-quarters full) helps prevent overfilling.
- Tamper-evident final closure — Once permanently sealed, the container cannot be reopened, preventing interference during storage and transportation.
MediWaste supplies a full range of UN-approved sharps containers specifically suited to dental practice requirements, including compact sizes ideal for bracket table mounting and larger options for oral surgery suites.
Safe Handling Procedures During Treatment
The majority of sharps injuries in dentistry occur during clinical procedures — not during disposal. Establishing safe handling procedures is therefore the single most impactful intervention for reducing injury risk. Every clinical protocol should incorporate sharps safety as a default consideration, not an afterthought.
The Prohibition on Two-Handed Recapping
The Health and Safety (Sharp Instruments in Healthcare) Regulations 2013 establish a strong presumption against the recapping of needles. Two-handed recapping — where one hand holds the needle cap while the other guides the needle into it — is the single greatest cause of preventable needlestick injuries in dentistry. It must never be performed under any circumstances.
Where recapping is deemed necessary (for example, between multiple injections during the same procedure), only a single-handed scoop technique or a proprietary recapping device may be used. The single-handed technique involves laying the cap on a flat surface, using the needle itself to scoop the cap onto the syringe, and then securing it against the surface. Purpose-built needle guards and recapping devices offer an even safer alternative.
Safer Sharps Devices
The regulations require that safer sharps devices are used wherever reasonably practicable. In dentistry, this includes:
- Safety-engineered dental syringes — These incorporate retractable needles or shielding mechanisms that activate automatically after injection, eliminating the exposed needle.
- Retractable scalpels — Disposable scalpels with blade retraction mechanisms that prevent post-use exposure to the cutting edge.
- Safety cannulae — For intravenous sedation, safety-engineered cannulae with automatic needle shields reduce the risk during venepuncture.
- Single-use biopsy punches — Replacing reusable punch biopsy instruments with single-use safety versions where available.
Safe Passing Techniques
The transfer of sharp instruments between operator and dental nurse is a high-risk moment. Safe passing protocols include:
- Neutral zone passing — Establishing a designated area (such as a specific zone on the bracket table) where instruments are placed down by one person and picked up by another, eliminating hand-to-hand transfer.
- Verbal communication — The operator should verbally announce when passing or requesting a sharp instrument, ensuring the receiver is prepared and attentive.
- Safe orientation — When direct passing is unavoidable, sharps should be passed handle-first with the sharp end pointed away from both parties.
- Eye contact confirmation — Both parties should confirm visual readiness before any transfer of sharp instruments.
Immediate Disposal Principles
The fundamental rule of sharps safety is: the user disposes. The person who uses or disassembles the sharp instrument is responsible for its immediate safe disposal. Sharps must never be:
- Left on work surfaces, bracket tables, or instrument trays for others to clear
- Passed to another person for disposal
- Placed in kidney dishes or other temporary receptacles
- Carried any distance to reach a sharps container (the container must be brought to the point of use)
- Bent, broken, or manipulated before disposal
Ensure Your Practice Has Safe, Compliant Sharps Disposal
MediWaste provides UN-approved sharps containers and scheduled collections tailored to dental practices of all sizes.
Free assessment - Tailored pricing - No obligation
The CQC Inspection Angle: What Inspectors Look For
Care Quality Commission inspections of dental practices include a thorough examination of sharps safety practices. Understanding what inspectors look for allows you to maintain inspection readiness at all times, rather than scrambling to address issues before an announced visit. CQC has published specific "mythbusters" relating to sharps in dental practice, making their expectations transparent.
Documentation Inspectors Will Request
- Sharps-specific risk assessment — A documented assessment that identifies all sharps used in the practice, evaluates the risks they present, and records the control measures in place. This must be reviewed annually or following any incident.
- Evidence of safer sharps consideration — Documentation showing that the practice has actively evaluated safer sharps devices, trialled them where appropriate, and recorded the rationale for selection or rejection of specific products.
- Sharps injury log — A complete record of all sharps injuries, including near-misses, with evidence of investigation and corrective actions taken.
- Training records — Evidence that all relevant staff have received sharps safety training, with dates, content covered, and refresher schedules documented.
- Waste transfer documentation — Consignment notes and waste transfer notes confirming that sharps waste is collected by an authorised carrier and sent to a permitted treatment facility.
- Standard operating procedures — Written protocols covering sharps use, disposal, injury response, and container management.
Physical Observations During Inspection
Beyond documentation, inspectors will physically observe your sharps management practices. They will check:
- That sharps containers are correctly assembled and labelled with the practice details and date of first use
- That containers are not overfilled beyond the three-quarters fill line
- That temporary closure mechanisms are used when containers are not in active use
- That containers are securely positioned and cannot be knocked over or accessed by unauthorised persons
- That no sharps are visible on work surfaces or in general waste streams
- That safer sharps devices are available and in use where applicable
- That staff can articulate the sharps injury procedure when asked
CQC Mythbuster Insight
CQC has explicitly stated that they do not mandate a specific brand of safer sharps device. What they require is evidence that the practice has considered the risks, evaluated available options, and made an informed decision. If conventional devices are retained for specific procedures, the rationale must be documented and defensible. The key is demonstrating a thoughtful, risk-based approach rather than simply ignoring the issue.
Sharps Injury Protocol: What to Do When a Needlestick Occurs
Despite the best preventive measures, sharps injuries can still occur. Having a clear, well-rehearsed response protocol is essential. Every member of the dental team must know exactly what to do in the event of a sharps injury, regardless of whether they are the injured party or a witness. Speed of response is critical — particularly regarding post-exposure prophylaxis for HIV, which is most effective when administered within one hour of exposure.
Immediate First Aid Actions
- Encourage bleeding — Gently squeeze the wound to encourage bleeding. Do not suck the wound, as this introduces oral flora and increases the area of exposure.
- Wash thoroughly — Clean the wound under running water with soap. Do not scrub aggressively, as this can cause tissue damage and potentially increase viral absorption.
- Apply a waterproof dressing — Cover the wound with an appropriate sterile dressing.
- If eyes or mouth are involved — Irrigate thoroughly with clean water or saline. Do not swallow any irrigation fluid.
- Do not use antiseptics or bleach — There is no evidence that applying antiseptic solutions reduces transmission risk, and they may cause tissue damage.
Reporting and Medical Assessment
- Report immediately — Notify the practice manager or designated person without delay. The injury must be recorded in the sharps injury log and the practice accident book.
- RIDDOR reporting — Consider whether the injury is reportable under the Reporting of Injuries, Diseases and Dangerous Occurrences Regulations 2013. Sharps injuries may be reportable as dangerous occurrences in certain circumstances.
- Urgent medical assessment — The injured person must attend the nearest Emergency Department or Occupational Health service as soon as possible for risk assessment and consideration of post-exposure prophylaxis (PEP).
- Source patient assessment — Where the source patient is known, they should be counselled and, with consent, tested for blood-borne viruses (Hepatitis B, Hepatitis C, and HIV). This information guides the medical management of the injured worker.
- Baseline blood samples — A blood sample from the injured person should be taken and stored (with consent) for retrospective testing if required.
Post-Exposure Prophylaxis
Post-exposure prophylaxis for HIV (PEP) involves a 28-day course of antiretroviral medication. It is most effective when started within one hour of exposure, retains some efficacy up to 72 hours, and is not recommended beyond this window. The decision to initiate PEP is made by the assessing clinician based on the nature of the injury, the source patient's risk factors, and their known or suspected HIV status.
For Hepatitis B, the response depends on the immunisation status of the injured person. Those with documented adequate antibody levels (above 100 mIU/ml) are generally considered protected. Those with uncertain or inadequate immunity may require Hepatitis B immunoglobulin and/or an accelerated vaccination course.
Critical Timeline
HIV post-exposure prophylaxis is most effective within ONE HOUR of exposure. Every dental practice must have a clear protocol identifying the nearest PEP provider and the fastest route to access treatment. This information should be prominently displayed and known to all clinical staff. Do not waste time with unnecessary internal procedures before seeking medical assessment.
Investigation and Learning
Every sharps injury, regardless of severity, must be investigated to determine its root cause and identify preventive measures. The investigation should consider:
- What task was being performed at the time of injury?
- Was the correct procedure being followed?
- Were safer sharps devices available and in use?
- Was the sharps container accessible and not overfilled?
- Were there environmental factors (poor lighting, rushing, interruptions)?
- What changes could prevent recurrence?
The findings of the investigation must be shared with the team (maintaining confidentiality regarding the injured person where appropriate) and used to update risk assessments and procedures as necessary.
Training Requirements for All Staff
Sharps safety training is not limited to clinical staff. The Health and Safety (Sharp Instruments in Healthcare) Regulations 2013 require that all workers who may be exposed to sharps receive appropriate training. In a dental practice, this potentially includes every member of the team.
Clinical Staff Training
Dentists, dental nurses, hygienists, and therapists require comprehensive training covering:
- Identification of all sharps used in the practice
- Correct assembly and use of safer sharps devices
- Single-handed recapping technique (where recapping is justified)
- Safe passing and neutral zone protocols
- Correct disposal procedures, including container assembly and closure
- Container management (fill levels, labelling, temporary closure)
- The sharps injury response protocol, including PEP access routes
- Reporting requirements and documentation
- Legal obligations under the 2013 Regulations
Non-Clinical Staff Training
Receptionists, practice managers, and cleaning staff also require sharps awareness training, albeit at a different level. They need to understand:
- What sharps containers look like and why they must never be touched or moved by unauthorised persons
- The immediate actions to take if they discover a sharp outside its container (for example, in general waste or on a surface)
- The sharps injury first aid procedure in case of accidental exposure
- Their reporting responsibilities if they witness unsafe practices or discover sharps in inappropriate locations
- The reason why sealed sharps containers awaiting collection must not be interfered with
Training Best Practice
Sharps safety training should be delivered at induction for all new staff, refreshed annually, and revisited immediately following any sharps injury or near-miss. Consider using scenario-based training that allows team members to practise the injury response protocol in a simulated environment. This builds muscle memory and reduces panic in real situations. Document all training with dates, attendees, content covered, and the trainer's details.
Competency Assessment
Training alone is insufficient without competency assessment. Practices should implement a system for verifying that staff can demonstrate safe sharps handling in practice. This might include direct observation of clinical procedures, questioning during appraisals, and periodic review of sharps disposal practice across all surgeries. Any deficiencies identified should trigger immediate retraining rather than waiting for the next scheduled session.
Storage and Collection Frequency
The management of sharps containers between filling and collection is an often-overlooked aspect of sharps safety. Correct storage practices protect against accidental injury, unauthorised access, and environmental contamination.
The Three-Quarters Rule
Sharps containers must be permanently sealed and replaced when they reach three-quarters full. This is not merely a guideline — it is a safety requirement that prevents injuries from protruding sharps when the container is being sealed or transported. Overfilling is one of the most common sharps safety violations identified during CQC inspections and is a frequent contributing factor in sharps injuries.
To ensure compliance with the three-quarters rule:
- Ensure all containers have a visible fill-level indicator line
- Train all staff to recognise when a container is approaching the maximum level
- Assign responsibility for monitoring container levels (typically to dental nurses in each surgery)
- Maintain a stock of replacement containers so that full containers can be immediately replaced
- Never attempt to compress or rearrange the contents of a container to create more space
Labelling Requirements
Every sharps container must be labelled with:
- The name and address of the practice (the waste producer)
- The date the container was first put into use
- The date the container was permanently sealed
- The name or initials of the person who sealed the container
This information provides traceability in the event of any incident during storage, collection, or treatment, and is a requirement under the duty of care regulations.
Storage Between Collection
Sealed sharps containers awaiting collection must be stored in a secure, designated area that is:
- Inaccessible to unauthorised persons, patients, and members of the public
- Protected from adverse weather conditions and extremes of temperature
- Away from food preparation or consumption areas
- Clearly signed as a clinical waste storage area
- Able to contain any spillage in the event of container failure
- Lockable and secure against vandalism or theft
Collection Frequency
The frequency of sharps collection depends on the volume of sharps generated by your practice and the available storage capacity. However, HTM 07-01 stipulates that sharps containers should not be stored for extended periods. As a general principle, sealed containers should be collected within a reasonable timeframe. Most dental practices find that monthly or fortnightly collections are appropriate, though high-volume practices may require weekly service.
MediWaste offers flexible dental waste collection schedules tailored to your practice's specific needs, ensuring that sealed containers are never stored longer than necessary.
Key Takeaway: The Complete Container Lifecycle
Every sharps container follows a defined lifecycle: assembly and labelling, placement at point of use, active filling with temporary closure between uses, monitoring of fill level, permanent sealing at three-quarters full, secure storage, and finally collection by an authorised waste carrier. Each stage has specific requirements, and failure at any point creates risk. Assign clear responsibilities for each stage and audit regularly.
Common Mistakes and How to Avoid Them
Even well-intentioned practices can fall into habits that compromise sharps safety. Based on our experience working with hundreds of dental practices across the United Kingdom, these are the most frequently observed errors and their solutions.
Mistake 1: Overfilling Sharps Containers
The problem: Attempting to maximise the use of each container by filling beyond the three-quarters line. This false economy creates a genuine injury risk when attempting to seal the container and during subsequent handling.
The solution: Treat the fill line as an absolute boundary. The cost of a replacement container is negligible compared to the cost of a sharps injury (estimated at over 500 pounds per incident when factoring in lost time, medical assessment, PEP consideration, follow-up testing, and administrative time).
Mistake 2: Using Containers Past Their Expiry
The problem: Sharps containers that have been in use for an extended period (particularly in low-volume rooms such as hygienist surgeries) may degrade, with lids becoming loose and materials becoming brittle.
The solution: Implement a maximum use period of three months from the date of assembly, regardless of fill level. When a container reaches three months old, seal it and replace it, even if it is not yet three-quarters full.
Mistake 3: Incorrect Container Positioning
The problem: Placing sharps containers on the floor, on high shelves requiring reaching, or in positions where the user must carry sharps any distance to reach them.
The solution: Wall-mount containers at a height between waist and shoulder level, within arm's reach of where sharps are used. If wall mounting is not possible, ensure the container is on a stable surface at an accessible height and cannot be knocked over.
Mistake 4: Disassembling Needles from Cartridges by Hand
The problem: Manually separating needles from anaesthetic cartridges or syringes using fingers, creating a high-risk exposure to the needle point.
The solution: Use the integral needle removal feature on the sharps container, or dispose of the complete assembly (needle and cartridge together) directly into the container. Many modern dental syringes allow the entire cartridge and needle assembly to be ejected directly without hand involvement.
Mistake 5: Failing to Use Temporary Closure
The problem: Leaving sharps containers fully open during sessions when they are not actively being used, creating a risk of spillage or inadvertent hand entry.
The solution: Engage the temporary closure mechanism between disposals. This takes only a second and significantly reduces the risk of accidental contact with container contents.
Mistake 6: Inadequate Segregation
The problem: Placing non-sharp clinical waste (cotton rolls, gauze, gloves) into sharps containers, or conversely, placing sharps into general clinical waste bags.
The solution: Sharps containers are exclusively for sharps. Non-sharp clinical waste goes into the appropriate waste stream (typically orange bags for treatment or yellow bags for incineration). Cross-contamination wastes expensive sharps container capacity and, more dangerously, placing sharps in soft clinical waste bags exposes waste handlers to needlestick injuries.
Mistake 7: No Near-Miss Reporting Culture
The problem: Only recording actual injuries while ignoring near-misses, which represent the majority of sharps safety events and provide crucial learning opportunities.
The solution: Establish a blame-free reporting system that actively encourages reporting of all near-misses. Analyse these reports for patterns and systemic issues. A practice that records zero near-misses is not a safe practice — it is a practice where staff do not feel safe reporting.
Building a Sharps Safety Culture in Your Practice
Technical measures and procedures are necessary but insufficient. True sharps safety requires a cultural commitment that permeates every level of the practice, from the principal dentist to the most junior team member. A strong safety culture means that everyone feels empowered to raise concerns, challenge unsafe practices, and contribute to continuous improvement.
Leadership Commitment
Safety culture starts at the top. Practice owners and principals must visibly demonstrate their commitment to sharps safety through their own behaviour, their investment in safety equipment, and their response to incidents and concerns. When leaders cut corners or dismiss concerns, the message cascades rapidly through the entire team.
Practical demonstrations of leadership commitment include:
- Investing in safer sharps devices even when conventional alternatives are cheaper
- Allocating adequate time for safe disposal rather than pressuring staff to rush between patients
- Responding positively and constructively to incident reports and concerns
- Participating personally in sharps safety training and audits
- Including sharps safety performance in team meetings and practice reviews
Empowering Staff to Speak Up
Every team member must feel confident that raising a sharps safety concern will be welcomed, not penalised. This means creating an environment where:
- Near-miss reports are treated as valuable intelligence, not evidence of incompetence
- Suggestions for safer working practices are actively sought and implemented where feasible
- Junior staff can challenge senior clinicians who are observed working unsafely
- Regular team discussions about sharps safety are normalised and constructive
- Anonymous reporting mechanisms are available for those who do not feel comfortable raising concerns openly
Regular Audit and Continuous Improvement
A systematic audit programme is essential for maintaining standards and driving improvement. Consider implementing:
- Monthly walkthrough audits — A designated person inspects all surgeries, checking container positioning, fill levels, labelling, and overall compliance with protocols.
- Quarterly sharps safety reviews — Analysis of all incidents, near-misses, and audit findings to identify trends and areas for improvement.
- Annual comprehensive review — Full reassessment of the sharps risk assessment, evaluation of new safer sharps devices available on the market, and review of all procedures and training materials.
- Benchmarking — Compare your injury rates and near-miss reports with available data from similar practices to contextualise your performance.
Peer Observation and Feedback
Formal peer observation programmes, where colleagues observe each other's sharps handling during routine procedures and provide constructive feedback, can be highly effective in identifying habitual unsafe behaviours that the individual may not recognise in themselves. These programmes work best when they are reciprocal, non-hierarchical, and focused on learning rather than judgement.
The Role of Your Waste Provider in Sharps Safety
Your clinical waste disposal partner plays a more significant role in sharps safety than simply collecting full containers. A good waste provider should be a proactive partner in your safety programme, offering expertise, support, and practical solutions that enhance your compliance and protect your team.
What to Expect from Your Provider
- Container supply — Your provider should supply UN-approved, BS 7320 compliant sharps containers in the sizes appropriate to your practice. They should offer guidance on sizing and positioning based on their experience with similar practices.
- Scheduled collections — Regular, reliable collection of sealed containers on an agreed schedule that prevents extended storage periods. The schedule should be reviewed periodically to ensure it remains appropriate for your waste volumes.
- Documentation — Provision of compliant consignment notes and waste transfer documentation, maintained in an accessible format and available for inspection or audit at any time.
- Compliance support — Guidance on regulatory changes, help with audit preparation, and advice on best practice. Your provider should be a source of expertise that complements your own knowledge.
- Emergency response — The ability to respond to urgent collection needs (for example, if a container is damaged or a spillage occurs) outside the normal schedule.
- Training support — Many providers offer training resources, guidance materials, and in some cases direct training delivery to support your team's competence.
MediWaste provides a comprehensive sharps management service for dental practices that encompasses all of these elements. Our team understands the specific challenges of dental sharps disposal and can tailor a service that meets your practice's unique requirements. Get a free quote to discover how we can support your sharps safety programme.
Choosing the Right Provider
When selecting or reviewing your sharps disposal provider, consider:
- Are they registered with the Environment Agency as an authorised waste carrier?
- Do they provide full documentation including consignment notes and duty of care transfer notes?
- Can they demonstrate that waste is sent to appropriately permitted treatment facilities?
- Do they supply containers that meet BS 7320 and UN3291 requirements?
- Is their collection schedule reliable and flexible enough to meet your needs?
- Do they offer support beyond simple collection (compliance advice, container supply, training)?
- Can they provide references from other dental practices they serve?
- Do they have appropriate insurance coverage?
Provider Partnership Tip
The cheapest waste disposal service is rarely the best value. A provider who helps you maintain compliance, avoid enforcement action, and prevent injuries delivers value far beyond the simple cost per collection. Consider the total cost of waste management, including the administrative burden, compliance risk, and the potential cost of incidents when comparing providers. Contact MediWaste to discuss how we can provide genuine partnership rather than simply a collection service.
Integrating Sharps Safety into Your Wider Infection Control Programme
Sharps safety does not exist in isolation. It is one component of a comprehensive infection prevention and control programme that protects patients, staff, and the wider community. Effective integration means that sharps safety considerations are embedded within every clinical protocol, every risk assessment, and every training programme rather than being treated as a separate, standalone topic.
Links to Other Infection Control Measures
Sharps safety intersects with numerous other infection control domains:
- Personal protective equipment — Gloves provide a degree of protection against superficial sharps contact but cannot prevent penetrating injuries. Heavy-duty gloves should be worn when handling sealed sharps containers during storage and preparation for collection.
- Immunisation — All clinical staff should be immunised against Hepatitis B with documented antibody levels confirming adequate protection. This is a fundamental prerequisite for anyone who may be exposed to sharps.
- Decontamination — Reusable sharp instruments (such as metal burs for autoclaving) must be safely handled during decontamination processes, with appropriate protocols for transport, cleaning, and inspection.
- Waste segregation — Sharps disposal is part of the broader waste management system. Staff who understand the complete waste segregation system are less likely to make errors with sharps specifically.
- Clinical governance — Sharps safety data (injuries, near-misses, audit results) should feed into the practice's broader clinical governance framework and be considered alongside other quality and safety indicators.
Technology and Innovation
The dental industry continues to evolve, with new technologies offering potential improvements in sharps safety:
- Computer-controlled local anaesthesia delivery systems — Devices such as The Wand reduce the need for conventional syringes and needles in certain applications.
- Needle-free injection systems — Jet injection technology that delivers anaesthesia without a conventional needle, eliminating the sharp entirely for surface anaesthesia.
- Laser dentistry — In some applications, lasers can replace scalpel blades for soft tissue procedures, removing the sharp from the equation.
- Digital impression taking — While not directly a sharps issue, the move from conventional to digital impressions removes one situation where sharp instruments (impression material syringes with metal tips) are used.
Practices should stay informed about these developments and consider adoption where they offer genuine safety benefits without compromising clinical outcomes.
Financial Considerations and Return on Investment
Investing in sharps safety delivers measurable financial returns beyond the obvious moral and legal imperatives. Understanding these returns can help justify expenditure on safer devices, training, and premium waste services to practice owners and managers.
The True Cost of a Sharps Injury
A single needlestick injury generates costs including:
- Immediate first aid and staff time for wound management
- Emergency Department or Occupational Health attendance (lost clinical time)
- Blood testing for the injured person (baseline and follow-up at 6 weeks, 12 weeks, and 6 months)
- Source patient counselling and testing
- Post-exposure prophylaxis medication if indicated (approximately 600 to 1000 pounds per course)
- Administrative time for incident investigation, reporting, and documentation
- Potential locum costs if the injured person requires time away from work
- Psychological impact on the injured person and potential counselling requirements
- Staff replacement and recruitment costs if the injured person leaves the profession
- Potential HSE investigation and enforcement costs
- Insurance premium increases
- Potential civil litigation and compensation claims
Conservative estimates place the total cost of a single sharps injury at between 500 and 2000 pounds for a straightforward case with no transmission, rising to potentially hundreds of thousands of pounds if seroconversion occurs. Against these figures, the additional cost of safer sharps devices, quality containers, and comprehensive training is minimal.
Conclusion: Sharps Safety as a Non-Negotiable Standard
Sharps disposal in dental practice is not a peripheral compliance issue to be addressed once and forgotten. It is a dynamic, ongoing commitment that requires vigilance, investment, and leadership. The consequences of failure — from blood-borne virus transmission to regulatory enforcement, from staff injury to practice closure — are too severe to tolerate a casual approach.
Every dental practice, regardless of size, specialty, or setting, must maintain:
- A comprehensive, current sharps risk assessment
- Documented evaluation and adoption of safer sharps devices
- UN-approved, correctly sized and positioned sharps containers in every treatment area
- Clear standard operating procedures for sharps use, disposal, and injury response
- Regular, documented training for all staff with competency assessment
- A robust incident reporting and investigation system covering injuries and near-misses
- A reliable waste partner providing compliant collection and full documentation
- A culture of safety where every team member feels empowered to challenge unsafe practices
The landscape of sharps safety continues to evolve as new devices, technologies, and guidance emerge. Practices that embed sharps safety into their daily culture rather than treating it as a periodic compliance exercise will be best positioned to protect their teams, satisfy regulators, and deliver the highest standards of care.
If you are uncertain whether your current sharps disposal arrangements meet the required standards, or if you are looking for a waste partner who truly understands the needs of dental practice, MediWaste is here to help. Our team of specialists can conduct a free assessment of your current arrangements, identify any areas for improvement, and provide a tailored solution that gives you complete confidence in your compliance.
Protect Your Team with Professional Sharps Management
From supply of UN-approved containers to safe collection and disposal, MediWaste handles the complete sharps lifecycle for dental practices.
Free site assessment - Tailored advice - No obligation
